Surgery for Acoustic Neuroma: Retrosigmoid Approach
This is a preview. Check to see if you have access to the full video. Check access
Resection of a Large Acoustic Neuroma through the Retrosigmoid Approach: Maximizing Efficiency and Facial Function
Please note the relevant information for patients with acoustic neuroma is presented in another chapter. Please click here for patient-related content.
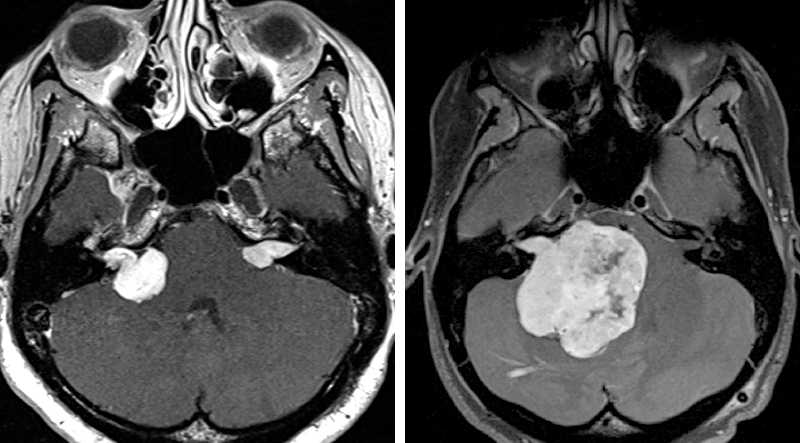
Vestibular schwannomas (VSs), also called acoustic neuromas, account for 8% to 10% of intracranial neoplasms. More specifically, they account for more than 80% of the cerebellopontine (CP) angle tumors, making them the most common tumor in this location. Most VSs arise from one of the vestibular nerves, most commonly from the superior one.
VSs are slow-growing benign neoplasms arising from the transition zone between the central and peripheral myelin, a point of origin typically found in the medial aspect of the internal auditory canal (IAC), approximately 8 to 12 mm from the pial surface of the pons. As they grow, VSs typically cause dysfunction of cranial nerve (CN) VIII, followed by dysfunction of CN VII. As the tumor reaches a giant size, it causes brainstem and cerebellar dysfunction.
Two genetic forms of VSs have been described: sporadic VSs and those associated with neurofibromatosis type 2 (NF2). Sporadic VSs are unilateral and account for about 95% of all VSs, whereas bilateral VSs are pathognomonic of NF2.
NF2 is a result of mutation in the NF2 gene that codes for Merlin, a tumor suppressor found on chromosome 22. Mutations in the NF2 gene have also been found in sporadic forms of VSs, as well as other tumors such as meningiomas. Attempts to distinguish sporadic from NF2-associated VSs based on specific NF2 gene mutations have not been successful, nor have attempts to correlate specific mutations with NF2 disease severity or aggressiveness of sporadic VSs.
Histologically, these different forms of VSs are indistinguishable, with the exception of a cystic subtype of sporadic VSs that harbors a higher degree of cellular atypia. This subtype tends to have a more aggressive clinical course and poorer surgical outcomes because it tends to be more adherent to the surrounding neurovascular structures.
VSs are classified based on their size because tumor size is the most important prognostic factor; 2 cm is an important threshold size above which outcomes have been shown to worsen.
Clinical Presentation
The most common presenting symptom of a patient with VS is gradual hearing loss, but occasionally hearing loss may be acute. Other common presenting symptoms include tinnitus and vertigo. Less common symptoms often indicative of large tumors include headache, facial numbness, ataxia, and nausea/vomiting.
Physical exam findings include unilateral sensorineural hearing loss. Large and giant tumors cause imbalance, abnormal corneal reflex, facial hypesthesia, nystagmus, and facial palsy. Abnormal extraocular muscle function and papilledema are less common and indicate the presence of hydrocephalus.
Evaluation
Otolaryngologic auditory testing assesses residual useful hearing and influences the choice of surgical approach. Pure-tone audiograms and speech discrimination testing are standard, commonly classified according to the Gardner-Robertson scale.
Auditory brainstem response testing can diagnose VSs, but it is less sensitive than magnetic resonance (MR) imaging, and therefore plays little role in the management of these tumors. The functions of the lower cranial nerves are formally established for large and giant tumors with inferior extension.
Diagnostic imaging includes a computed tomography (CT) scan to evaluate the bony anatomy that will be removed during creation of the surgical corridor. Recognition of the extent of pneumatization of the temporal bones and appropriate preparation for intraoperative repair can minimize the risk of a postoperative cerebrospinal fluid (CSF) leak. The position of the venous anatomy, specifically a high-riding jugular bulb or posteriorly displaced sigmoid sinus, may affect the choice of the operative corridor and the extent of bone removal. Tumor anatomy and its approximate relation to the nearby neurovascular structures are determined with MR imaging.
The relationship of the tumor to the basilar and posterior inferior cerebellar arteries may be appreciated; such vessels should be carefully protected if ultrasonic aspirator devices are used during surgery for tumor decompression. Despite extension of the large and giant tumors through the jugular foramen, they are microsurgically dissected off of the lower cranial nerves without significant risk.
The presence of edema in the brainstem indicates a high risk of brainstem pial violation during microdissection of the tumor capsule. If significant brainstem edema is evident, staging the surgery may be considered because the interval between the two stages allows the tumor to deliver itself into the resection cavity created during the first operative session. Subtotal removal is an alternative option to avoid brainstem injury.
VSs may be occasionally difficult to distinguish from CP angle meningiomas on MR imaging. Both are often homogeneously enhancing, but meningiomas are typically centered away from the IAC and have a broad dural attachment (tail) to the posterior aspect of the petrous ridge or tentorium. Meningiomas rarely originate in the IAC, and although they can spread secondarily into the IAC, the IAC is usually not widened. The angle between the tumor and the petrous pyramid is wide.
Centered around a widened IAC, VSs form an acute angle with the petrous pyramid and are likely to have a cystic component and be partially surrounded by a CSF cleft. The T2 sequences determine pial invasion (brainstem edema) or encasement of the vasculature.
The majority of patients do not require treatment for their hydrocephalus beyond tumor resection; however, a minority require postoperative ventriculoperitoneal shunting.
As expected, increased tumor size is a risk factor for the development of postoperative hydrocephalus. If symptomatic obstructive hydrocephalus is present, I prefer to place an external ventricular drain (EVD) at the time of the craniotomy with the goal of weaning the patient off the drain postoperatively or shunting if necessary.
Indications for Surgery and Treatment Options
Management options for VSs include observation with serial imaging, microsurgical resection, stereotactic radiosurgery (SRS), and a combination of resection and SRS. Small asymptomatic tumors or minimally symptomatic tumors in older patients and patients with high surgical risk may be observed with repeat MR imaging.
The majority of VSs will continue to grow, but their natural history is unpredictable, with some tumors remaining unchanged or rarely even regressing over time. Treatment is indicated for progressively enlarging tumors. If the tumor is small (<3 cm), SRS is a reasonable option that has demonstrated excellent tumor control with low treatment-related morbidity.
Patients harboring recurrent or residual tumors are referred for radiosurgery rather than reoperation. Outcomes for facial nerve function and hearing are more favorable with radiosurgery than with traditional open surgery. Radiosurgery has recently been advocated as the primary therapy for larger tumors, but reliable data currently do not exist about the outcomes of radiosurgery for this specific population
Tumors causing symptoms or neurologic dysfunction secondary to mass effect are surgically resected with the goal of radical subtotal removal to preserve facial function. This form of resection supplemented by SRS for a growing residual tumor is an emerging paradigm for the treatment of skull base tumors.
In summary, I have a low threshold for the use of radiosurgery in the setting of small and medium sized tumors (<3cm) if the mass effect is not considered prohibitive for radiosurgery. Very young patients with medium size tumors fair better from microsurgery long-term. Large and giant tumors deserve surgical removal. Patients with NF2 present a daunting challenge in their management as the goal is to protect some hearing long-term; their care has to be individualized.
Preoperative Considerations
Routine use of intraoperative facial nerve electromyography (EMG) and brainstem auditory evoked response potentials (BAERs) monitoring to identify and monitor facial nerve and cochlear nerve/brainstem integrity, respectively, is important. The significant attenuation and atrophy of the facial nerve in giant tumors, as apparent from preoperative facial weakness, often complicates mapping the exact location of the nerve splayed over the tumor capsule.
Staging the operative session should be strongly considered and preplanned for giant tumors. Fatigue of the surgeon and the operative team during the later (and more critical) parts of the operation is an important factor in poor outcomes.
A high-riding jugular bulb close to the IAC should be recognized on preoperative imaging and the surgeon should be cognizant of this anatomic finding when drilling the IAC.
Operative Anatomy
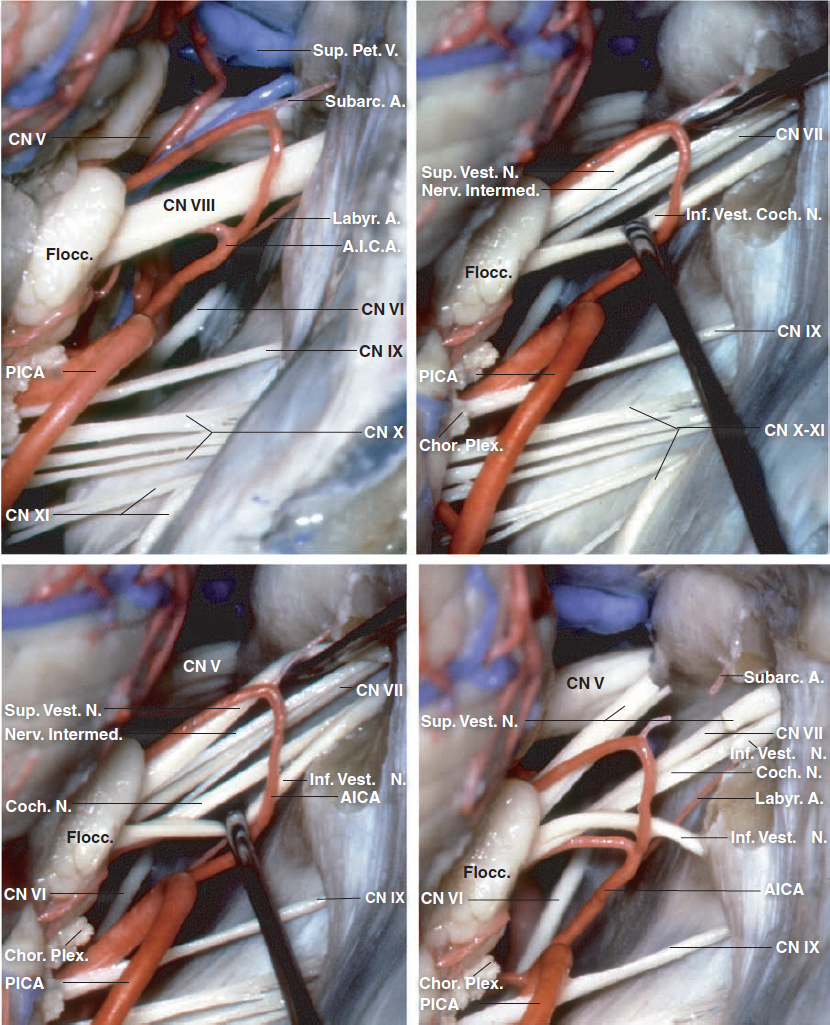
The neural anatomy of the posterior fossa, especially near the porus acousticus, is relevant to microsurgery of VSs.
Click here to view the interactive module and related content for this image.
Figure 2: The cranial nerves of the CP angle through a right-sided retrosigmoid approach are shown. The vestibulocochlear nerve enters the IAC along with a labyrinthine branch of the anterior inferior cerebellar artery (AICA). The posterior wall of the IAC has been removed in the last three images to show the cleavage plane between the upper components of CN VII/VIII complex formed by the superior vestibular nerve and facial nerve, and the lower components formed by the inferior vestibular and cochlear nerves. The facial nerve is located anterior to the superior vestibular nerve and the cochlear nerve is anterior to the inferior vestibular nerve. The superior vestibular nerve is posterior and superior, the facial nerve anterior and superior, the inferior vestibular nerve posterior and inferior, and the cochlear nerve anterior and inferior (images courtesy of AL Rhoton, Jr).
Click here to view the interactive module and related content for this image.
Figure 3: The posterior wall of the IAC has been resected to show the neural anatomy within the canal. The transverse crest separates the superior vestibular and facial nerves above from the inferior vestibular and cochlear nerves below. The separation plane between the superior and inferior vestibular and cochlear nerves has been exaggerated to expose the individual nerve bundles (images courtesy of AL Rhoton, Jr).
What is the Best Surgical Approach in VS Surgery?
VSs have been resected via the translabyrinthine (TL), middle cranial fossa (MCF), and retrosigmoid (RS) approaches, with the choice of approach based on the size/location of the bulk of the tumor, status of the patient’s hearing, and the preference of the operating surgeon. Other factors that play a role in selection of the approach include the patient’s age and overall health status, the anatomy of the vestibule and cerebellopontine (CP) angle, and involvement of the brainstem and the internal auditory canal (IAC). The RS approach is favored by neurosurgeons and offers an opportunity for hearing preservation while tackling large tumors via a panoramic view of the CP angle.
VSs have been stratified into the following tumor size/location categories: intracanalicular (IC), <1.5 cm (small), 1.5 to 3.0 cm (medium), and >3.0 cm (large). The MCF option may be superior to RS surgery for hearing preservation in patients with tumors <1.5 cm. Some neurosurgeons argue that the RS corridor results in less neurologic dysfunction during resection of the IC tumors than the MCF route. The RS pathway is potentially associated with less dysfunction than the MCF or TL approaches for tumors that are 1.5 to 3.0 cm.
Postoperative headaches tend to be more prevalent after the RS than TL route. The risk of CSF leak is greater following the RS craniotomy than with either the MCF and TL approaches. Some investigators have reported that the incidences of mortality, major noncranial nerve neurologic complications, residual tumor, tumor recurrence, and dysfunction of other CNs are not significantly different with the various approaches. There are no class I data that strongly support any one approach over another.
In summary, the MCF route appears to be safest for hearing preservation among smaller tumors. The RS trajectory is potentially the most versatile corridor for most tumor sizes, but it is associated with a higher risk of postoperative pain and CSF fistula. Finally, the TL osteotomy is associated with complete hearing loss, but may be beneficial for patients with large tumors and poor preoperative hearing.
Venous anatomy also plays a role in selection of the surgical approach. A high-riding or anteriorly located sigmoid sinus favors the RS over the TL route. Younger patients with “full” cerebellum and large tumors can benefit from the TL osteotomy because there is less need for cerebellar mobilization and retraction during this approach.
Opponents of RS surgery quote cerebellar retraction as a risk for postoperative ataxia and maintain that this approach carries a higher incidence of postoperative headache. This approach may also provide only a limited visualization of the fundus of the IAC, which may necessitate blind dissection to remove the entire tumor. However, the RS approach is a versatile route for tumors of any size regardless of the patient’s preoperative hearing status.
In the modern era, when the goal of zero mortality and minimal morbidity is achievable, hearing conservation should be pursued; however, with large and giant tumors, hearing preservation is the exception rather than the rule.
RESECTION OF VESTIBULAR SCHWANNOMA VIA A RETROSIGMOID CRANIOTOMY
For moderate size tumors, I place a lumbar drain to facilitate relaxation of the cerebellum during the dural opening. For very large tumors causing significant obstructive hydrocephalus, an external ventricular drain is installed. This early release of CSF avoids cerebellar herniation that commonly occurs because of the mass effect of VSs.
The patient may be positioned in the sitting/semisitting or horizontal (supine, oblique, park bench) positions. The sitting position is associated with a risk of venous air embolism; precordial Doppler and end-tidal CO2 monitoring are used.
Some operators report less blood loss, shorter operative time, and lower risk of cranial nerve dysfunction with the sitting position. This position facilitates a clear surgical field via gravity drainage of blood and CSF. The use of suction device and bipolar coagulation are therefore minimized around the cranial nerves. In addition, atraumatic bimanual microdissection can be accomplished using two microforceps to disconnect the fine arachnoid bands between the tumor and the encasing sheaths of the nerves and pia.
Bimanual microdissection provides a great advantage without the use of bipolar electrocautery that can place the facial nerve at risk. However, the sitting position requires nonergonomic posture of the surgeon’s arm, which leads to early fatigue. Most anesthesiologists are not comfortable with the sitting position, and therefore its use is limited in the United States.
I avoid the supine position due to the risk of neck stiffness associated with long operative sessions. I prefer the park-bench position. For technical nuances of patient positioning, exposure and dural opening, refer to the chapter on Extended Retrosigmoid Craniotomy.
Figure 4: The curvilinear incision for medium to large size acoustic neuromas has a broader base than the one used for the standard retromastoid craniotomy aimed at microvascular decompression surgery. The sagittal suture is parallel to the surface of the floor. The shoulder is mobilized anteroinferiorly.
Figure 5: Note the generous retrosigmoid craniotomy for an acoustic neuroma (AN) in comparison with those for trigeminal neuralgia (TN) and hemifacial spasm (HS). Giant tumors demand a generous exposure to deal with the potential occurrence of cerebellar swelling.
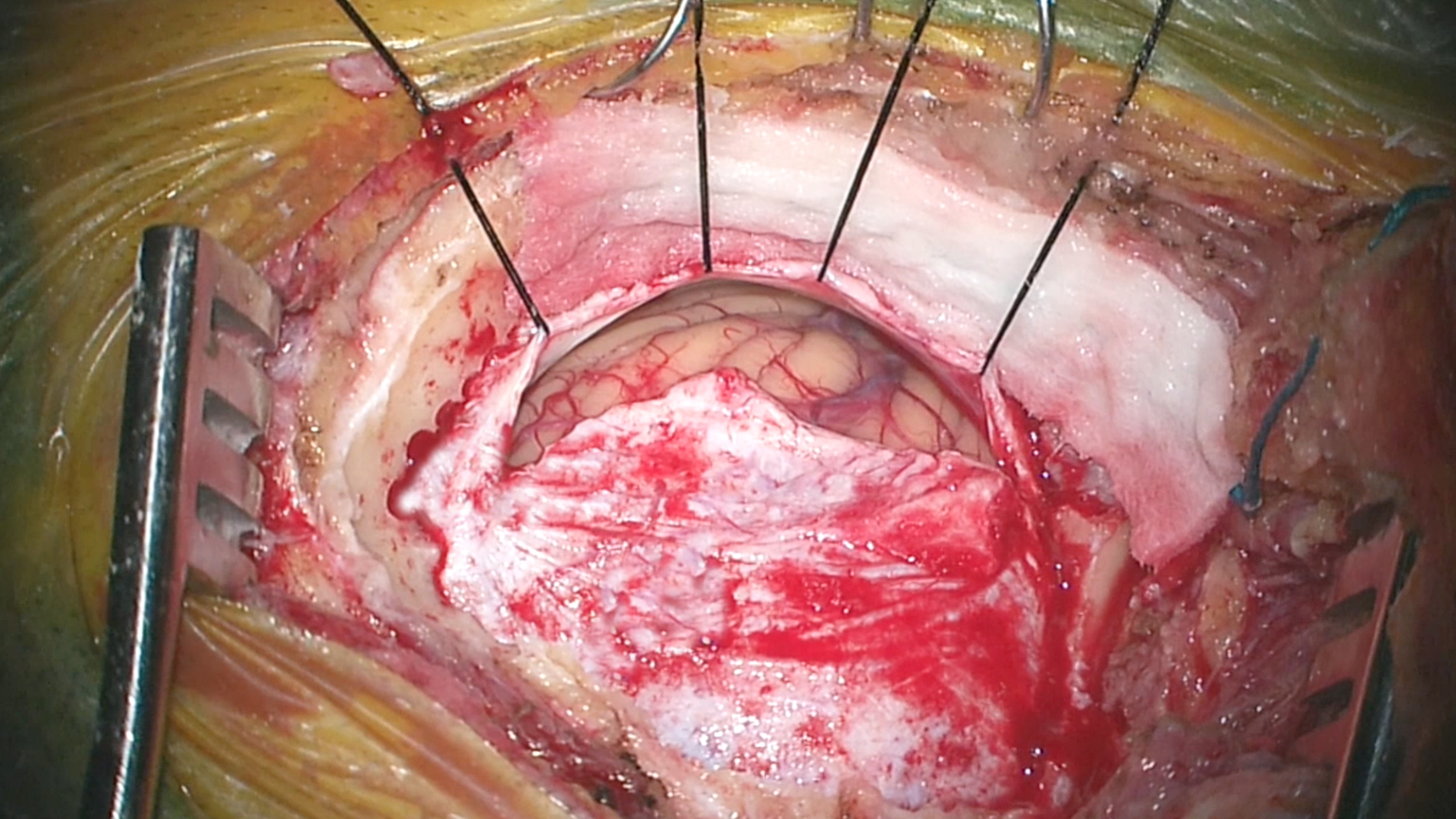
A large retromastoid craniotomy/ectomy extends along the edges of the transverse and sigmoid sinuses. Smaller craniotomy/ectomy may not provide adequate decompression if intraoperative cerebellar swelling is encountered. Bone removal for large tumors extends to the level of the posterior fossa floor, but does not involve unroofing the foramen magnum.
Figure 6: The dura is incised along the transverse and sigmoid dural venous sinuses. This method of dural opening leaves most of the dura on the cerebellum, covered by the cottonoid patties, and protects the dura from desiccation/shrinkage caused by the heat from the intense light of the microscope. Skeletonization of the dural venous sinuses will mobilize these sinuses via the retraction dural sutures and allows a more expanded operative trajectory around the cerebellum.
INTRADURAL PROCEDURE
The petrous-tentorial junction is identified to orient the surgeon regarding the route of the intradural trajectory. Inadvertent supracerebellar dissection can lead to avulsion of the bridging veins and torrential venous bleeding.
Figure 7: Additional CSF is released by opening the arachnoid membranes caudal to the tumor and over the lower cranial nerves (inset image). Careful management of cerebellar swelling via medical (mannitol, diuretics) and surgical (CSF drainage) means is important to avoid undue cerebellar retraction that exacerbates swelling. If possible, the superior petrosal vein is protected during the early stages until substantial tumor debulking is accomplished. This maneuver minimizes the risk of cerebellar venous congestion.
A small lateral portion of the cerebellum may be excised to allow adequate tumor exposure without aggressive cerebellar retraction (this maneuver may be necessary only in younger patients with a “full” cerebellum, otherwise cisternal opening and CSF drainage is adequate). Next, the arachnoid bands over the posterior aspect of the tumor capsule are released. Stimulation of the posterior/inferior surface of the capsule will exclude an aberrant posterior/inferior location of the facial nerve.
Figure 8: A monopolar stimulator probe maps the posterior and inferior surfaces of the capsule to exclude the presence of CN VII and allows a generous incision within the capsule in expectation of aggressive intracapsular debulking. I start my stimulation threshold at 0.1 mA, and if I find a response in an area, the signal is decreased to 0.07mA for more refined mapping.
Figure 9: Maximal safe internal tumor debulking using an ultrasonic aspirator without interruption of the anterior, superior, and inferior poles/capsule of the tumor is the key maneuver and tremendously facilitates the later stages of the operation. This technique provides enhanced tumor capsule dissection and mobilization while decreasing neural tissue stretch/retraction.
Following devascularization of the tumor by coagulating the feeders from the dura over the porus acusticus, microdissection proceeds inferiorly. If the tumor has substantial inferior extension, the enucleated capsule is mobilized away from the lower cranial nerves and out of the jugular foramen. The decompressed tumor is mobilized away from the nerves rather than vice versa.
Figure 10: Removal of tumor from the jugular foramen and lower cranial nerves should be atraumatic because the lower cranial nerves do not significantly attach to the capsule. Before manipulation of the lower pole, stimulation of the capsule excludes the presence of the facial nerve. Note that the nerve may have an undulating course and reach the inferior pole just before it enters the porus. The encasing arachnoid layers of the nerves are used as a “handle” by the microforceps to mobilize the nerves. Direct handling of the nerves is avoided.
Once the inferior pole is resected and shrunken, the tumor is internally debulked further along its superior pole and rostral dissection proceeds, mobilizing the capsule away from the tentorium and CN V. The facial nerve may be draped over this pole and adjacent to CN V. This portion of the capsule should be carefully mapped with the stimulator probe before its dissection.
Contraction of the temporalis muscle caused by stimulation of the motor fibers of the trigeminal nerve should not be mistaken for localization of the facial nerve. Mapping of an attenuated/atrophied facial nerve in a patient with a giant tumor can be difficult, especially in the presence of preoperative facial weakness. Repetitive mapping at slightly higher stimulation parameters may be necessary to completely exclude the presence of the facial nerve in the region.
The capsule is sharply dissected away from the trigeminal nerve root entry zone and is rolled from the medial to lateral direction. The trochlear nerve and superior cerebellar artery are usually encased within thick arachnoid bands and are readily mobilized.
Figure 11: Dissection of the tumor away from CN V is shown. The superior petrosal vein is protected if possible. The lower cranial nerves are covered with a small piece of cottonoid. The lower intraoperative photo shows the root entry zone of CN V.
Next, the thoroughly cored out tumor capsule is rolled laterally away from the middle cerebellar peduncle and brainstem toward the porus. Meticulous hemostasis will allow the operator to appreciate the most important factor in safe resection of these challenging tumors: microdissection along the arachnoid membranes and protection of the brainstem pial surfaces.
The capsule is gently pulled upon in the lateral direction via tumor forceps as extra-arachnoid dissection is performed using microforceps to detach the arachnoid membranes from the tumor. Periodic irrigation by the assistant clears the field.
Direct placement of the suction device over the brainstem and cranial nerves is avoided. The veins over the surface of the brainstem are engorged and prone to avulsion, leading to blood loss and interference with adequate visualization of the dissection planes. Gentle pressure/tamponade using a cotton ball over the site of the hemorrhage followed by coagulation of the vein along its more proximal segment away from the brainstem is a reasonable strategy.
Again, stepwise strategic internal tumor debulking using an ultrasonic aspirator followed by tumor mobilization is an efficient and safe maneuver to avoid inadvertent injury by undue traction on the surrounding structures.
Further mobilization of the inferior pole of the tumor should protect the posterior inferior cerebellar artery and its branches. En passage vessels are microsurgically mobilized using sharp dissection, while small tumor-feeding vessels are carefully coagulated and cut. Blunt dissection of the perforators and their subsequent avulsion is avoided.
CN VIII is encountered during dissection of the inferior pole of the tumor from the brainstem. Stimulation mapping avoids inadvertent injury to CN VII that may be near CN VIII. Preservation of CN VIII in patients with large and giant tumors is almost impossible and not advisable if preoperative hearing was deemed nonfunctional. This nerve is isolated and sharply cut in large tumors. Inadvertent traction on the nerve is prevented.
In the presence of preoperative middle cerebellar peduncle and/or brainstem edema, violation of the pial membranes is likely during resection of large and giant tumors. If such violation occurs, a small piece of cottonoid may be used to mobilize (peel away/”wipe away”) the brainstem from the tumor capsule without placing the former at risk of injury by the suction apparatus. Most of the edema is usually within the middle cerebellar peduncle.
Figure 12: Cottonoid patties are used to “wipe” the middle cerebellar peduncle away from the mobilized tumor capsule where the pial planes are not preserved (top sketch). If the pial planes are not recognizable at one point, the dissection is diverted to another location (the poles of the tumor) where the planes are more recognizable. If the arachnoid planes are intact (lower photo), the microforceps are used to mobilize the pia away from the tumor. The small pial vessels are protected when possible and coagulation minimized. Pial bleeding is controlled via gentle tamponade using thrombin-soaked Gelfoam pledgets and irrigation fluid.
As the tumor is mobilized away from the brainstem along its superior pole and midsection, stimulation mapping will localize the facial nerve at the tumor capsule or at its exit zone near the brainstem. The most reliable maneuver to expose the nerve safely is to peel the tumor laterally and identify the nerve at its root exit zone near the brainstem.
Removal of the tumor at the depth of the cerebellopontine cleft/fissure may require the greatest amount of cerebellar retraction; changing the angle of the microscope’s view and intermittent dynamic retraction using the handheld suction device will minimize the required persistent force on the neural structures. During this phase of the operation, any changes in vital signs, significant violation of the pial membranes, or worrisome facial nerve recordings should lead the operator to adjust his or her operative strategy or even consider staging the operation.
After localization of the facial nerve along the superior half of the capsule, I remove the inferior half of the tumor guided by stimulation mapping. CN VI may be adherent to a giant tumor.
As the superior pole of the tumor is peeled off, I expose the root entry zone of CN V thoroughly. This part of the nerve is adherent; meticulous dissection will minimize the risk of postoperative trigeminal neuropathy and resultant corneal anesthesia.
Figure 13: Sharp dissection techniques and attentive internal decompression of the residual tumor will facilitate mobilization of the capsule. The nerve is stimulated at 0.07 mA along its length so that its path on the tumor capsule can be tracked. I place gentle tension on the tumor capsule and then peel the nerve away from the tumor (top image). CN VIII is identified and sacrificed in patients with larger tumors after its stimulation mapping (bottom image).
Figure 14: The suction device places the tumor capsule under gentle tension while the nerve is carefully mobilized away from the capsule. Preservation of CN VIII is possible in patients with medium size tumors (top image). Intensive tumor debulking and meticulous hemostasis are hallmarks of efficient VS surgery (bottom image) while the pial planes are preserved.
Bleeding can be a nuisance, but blind bipolar coagulation should be avoided. Some minor oozing is tolerated as long as microdissection can proceed with reasonable visualization of the operative field.
At this stage of the procedure, I take a break while my neuro-otology colleague removes the tumor within the IAC.
Removal of the Intracanalicular Component of the Tumor
The inferior wall of the IAC is drilled to the level of the fundus after the dura is dissected from the petrous bone. Cutting and diamond burrs remove the bone using generous irrigation fluid to avoid heat injury to the nerves within the canal. A large piece of Telfa is used to cover the cerebrovascular structures and the tumor within the CP cisterns to prevent bone dust from covering the operative field.
Ultimately, bone removal allows identification of the distal segments of CNs VII/VIII complex free of the tumor within the canal. Air cells are often entered and should be carefully waxed at the end of this stage.
Figure 17: The tip of the stimulator probe maps the normal segment of CN VII within the distal canal and anterior to CN VIII (Modified and Redrawn from Tew, van Loveren, Keller*).
Figure 18: The intracanalicular component of the tumor is debulked and rolled medially. The facial nerve is reidentified via mapping, and the vestibular nerve is sectioned to allow the tumor to be mobilized out of the canal (Modified and Redrawn from Tew, van Loveren, Keller*).
The facial nerve becomes attenuated and most adherent to the tumor along the junction of its cisternal and intracanalicular segments at the porus. The operator must use a combination of gentle blunt and sharp microdissection techniques within both the cisternal and canalicular planes to avoid facial nerve injury.
Removal of the tumor within the IAC and exposure of the facial nerve at the porus will provide important anatomic information regarding the turn of the nerve around the tumor capsule at the porus and the route of the facial nerve. This information can estimate the course of the nerve from its already identified root exit zone near the brainstem to the area of the porus. If the facial nerve is significantly attenuated, its anatomic preservation may not be possible in patients with giant tumors.
Gross total tumor resection is attempted. However, if the tumor is very adherent to the facial nerve near the porus, I leave a small sheet of the tumor over the nerve to optimize facial function. This residual small piece of the tumor can be managed postoperatively through surveillance imaging and treated with radiosurgery if its growth is detected.
The surgeon may use the blunt-tipped stimulator as a dissecting instrument to peel away the nerve from the residual capsule. Significant tension on the nerve is avoided and sharp dissection is used when possible. Shear injury to the nerve as a result of excessive traction is irreversible.
Figure 19: The capsule is dissected away from the nerves. Any injury to the facial nerve requires an increase in the stimulation intensity to map the nerve. This finding indicates the need to avoid further dissection of the nerve and plan a radical subtotal removal of the tumor. The labyrinthine artery is preserved if CN VIII has remained intact. The arachnoid membranes over CN VII are left intact (Modified and Redrawn from Tew, van Loveren, Keller*).
Figure 20: The final resection result is illustrated. The typical course of the CN VII/VIII complex around the tumor is apparent (Modified and Redrawn from Tew, van Loveren, Keller*).
The anterior aspect of the tumor capsule contains small arteries and, more commonly, veins near the facial nerve. Bleeding from these vessels can be a significant source of annoyance. Gentle irrigation, application of tamponade using pieces of thrombin-soaked cotton, and patience are needed to avoid aggressive coagulation and suction that can easily injure the nerve.
After the tumor has been completely removed or subtotally resected, the surgical field is inspected. Response of the facial nerve to stimulation of 0.07 mA at its exit zone near the brainstem is a good indicator of acceptable facial function after surgery.
Figure 21: Radical subtotal tumor removal allows preservation of facial function. The small residual tumor that was intimately attached to the nerve at the porus was left behind (T).
Closure
The airs cells at the porus are rewaxed. Meticulous hemostasis is followed by watertight dural closure and generous application of bone wax to the mastoid air cells. If cerebellar swelling is apparent, the bone flap should not be replaced and a suboccipital decompressive craniectomy and duraplasty are performed.
Additional Considerations
A number of different facial nerve pathways around the tumor capsule have been described. These pathways are summarized below.
Figures 22: Various patterns of CN VII displacement around large and giant tumors are illustrated in the order of their incidence. The location of CN VIII is variable and not known in patients with larger tumors due to its significant attenuation.
Additional Considerations
Resection of small VSs presents less technical challenge.
Figure 23: A right-sided 1.7-cm VS was removed via the retromastoid approach. Note the relationship of the tumor to CN VIII (second photo). The bottom two photos demonstrate resection of the intracanalicular and intracisternal components of the tumor. The facial nerve was dissected free of the tumor capsule in the bottom photo.
Figure 24: Different dissection planes along different segments of the facial nerve are demonstrated. The top photo shows ease of dissection close to the brainstem. The middle one indicates the increased difficulty of dissection along the cisternal segment and finally the last photo discloses the tedious dissection of the attenuated nerve at the level of the porus.
Resection of an Intracanalicular Acoustic Neuroma
Postoperative Considerations
After surgery, patients are observed in the intensive care unit for any signs of neurologic deterioration, sudden hypertension, or ventilatory difficulty. Appropriate eye care is provided. The combination of trigeminal neuropathy and facial nerve paralysis places the eye at significant risk and appropriate precautions are taken to minimize the risk of corneal ulceration.
If dysphagia or respiratory insufficiency is encountered, early percutaneous gastrostomy and tracheostomy tubes are necessary to avoid aspiration pneumonia and hypoxemia.
The most feared complications are intracerebellar hemorrhage and progressive edema leading to symptomatic brainstem compression. Avoidance of significant cerebellar retraction, meticulous microdissection along the arachnoid and pial membranes, and prompt management of postoperative hypertension will minimize these unfortunate events.
Watertight dural closure minimizes the risk of pseudomeningocele formation. If rhinorrhea is encountered, I ask my otology colleagues to perform a mastoidectomy and obliterate the air cells with fat graft. In other words, the operative field is not re-explored.
The main neurologic morbidity of acoustic neuroma surgery is facial nerve palsy, which is both functionally and psychologically damaging to the patient and should be avoided through radical subtotal tumor removal. The size of the tumor is the main predictive factor of postoperative facial weakness.
Despite anatomic preservation of the facial nerve, some patients suffer complete facial paralysis after surgery. These patients should be reassured that reasonable recovery of facial function is possible. In the first few days after surgery, the eye is covered with a protective shield and viscous eyedrops are prescribed. Shortly thereafter, gold weight placement within the eyelid and/or tarsorrhaphy are performed. The patient is asked to protect the eye and wear protective spectacles.
At least 6 months of expectant management for the facial nerve palsy is provided before any further treatment is recommended. If permanent facial paralysis is evident, a faciohypoglossal anastomosis is recommended after the first postoperative year. This maneuver is also considered earlier if the nerve was anatomically noncontinuous at the conclusion of surgery.
If a staged operation is planned, the interval between operations may be 2 to 4 weeks, based on the patient’s recovery process from the first operation. Some surgeons advocate for a longer interval to allow further recovery of the cranial nerves and brainstem. If poor facial function is present from the first operation, the second stage is delayed until the nerve achieves a good functional recovery so it can be monitored via stimulation mapping during the second stage.
Pearls and Pitfalls
- Radical subtotal tumor resection is advised to avoid facial nerve palsy. The results of radiosurgery for treatment of growing residual tumors are very encouraging.
- Peeling the tumor from the medial to lateral direction has the best chance of protecting the facial nerve.
- Operative efficiency avoids surgeon’s fatigue and leads to improved outcomes.
Contributor: Andrew R. Conger, MD, MS
*Redrawn with permission from Tew JM, van Loveren HR, Keller JT. Atlas of Operative Microneurosurgery, WB Saunders, 2001. © Mayfield Clinic
For additional illustrations of facial nerve anatomy, please refer to the Jackler Atlas by clicking on the image below:
For additional illustrations of facial nerve repair, please refer to the Jackler Atlas by clicking on the image below:
For additional illustrations of the hypoglossal-facial anastomosis, please refer to the Jackler Atlas by clicking on the image below:
For additional illustrations of a retrosigmoid approach to the internal auditory canal and cerebellopontine angle, please refer to the Jackler Atlas by clicking on the image below:
For additional illustrations of tumor growth patterns, please refer to the Jackler Atlas by clicking on the image below:
DOI: https://doi.org/10.18791/nsatlas.v5.ch08.1
Some of the material included in this chapter was included in the following articles:
Ansari SF, Terry C, Cohen-Gadol AA. Surgery for vestibular schwannomas: a systematic review of complications by approach. Neurosurg Focus. 2012;33:E14.
Kulwin CG, Cohen-Gadol AA. Technical nuances of resection of giant (> 5 cm) vestibular schwannomas: Pearls for success. Neurosurg Focus. 2012;33:E15.
References
Black S, Ockert DB, Oliver WC Jr, Cucchiara RF. Outcome following posterior fossa craniectomy in patients in the sitting or horizontal positions. Anesthesiology 1988;69:49-56.
Briggs RJ, Shelton C, Kwartler JA, Hitselberger W. Management of hydrocephalus resulting from acoustic neuromas. Otolaryngol Head Neck Surg. 1993;1020-1024.
Charpiot A, Tringali S, Zaouche S, Ferber-Viart C, Dubreuil C. Perioperative complications after translabyrinthine removal of large or giant vestibular schwannoma: outcomes for 123 patients. Acta Otolaryngol. 2010;130:1249-1255.
Comey CH, Jannetta PJ, Sheptak PE, Joh HD, Burkhart LE. Staged removal of acoustic tumors: techniques and lessons learned from a series of 83 patients. Neurosurgery. 1995;37:915-920; discussion 920-911.
Duke DA, Lynch JJ, Harner SG, Faust RJ, Ebersold MJ. Venous air embolism in sitting and supine patients undergoing vestibular schwannoma resection. Neurosurgery. 1998;42:1282-1286; discussion 1286-1287.
Elsmore AJ, Mendoza ND. The operative learning curve for vestibular schwannoma excision via the retrosigmoid approach. Br J Neurosurg. 2002;16:448-455.
Falcioni M, Fois P, Taibah A, Sanna M. Facial nerve function after vestibular schwannoma surgery. J Neurosurg. 2011;115:820-826.
Fundova P, Charabi S, Tos M, Thomsen J. Cystic vestibular schwannoma: surgical outcome. J Laryngol Otol. 2000;114:935-939.
Gardner G, Robertson JH. Hearing preservation in unilateral acoustic neuroma surgery. Ann Otol Rhinol Laryngol. 1988;97:55-66.
Gerganov VM, Pirayesh A, Nouri M, Hore N, Luedemann WO, Oi S, et al. Hydrocephalus associated with vestibular schwannomas: management options and factors predicting the outcome. J Neurosurg. 2011;114:1209-1215.
Gormley WB, Sekhar LN, Wright DC, Kamerer D, Schessel D. Acoustic neuromas: results of current surgical management. Neurosurgery. 1997;41:50-58; discussion 58-60.
Jackler RK, Pitts LH. Acoustic neuroma. Neurosurg Clin N Am. 1990;1:199-223.
Jackler RK, Pitts LH. Selection of surgical approach to acoustic neuroma. 1992. Neurosurg Clin N Am. 2008;19:217-238.
Mamikoglu B, Wiet RJ, Esquivel CR. Translabyrinthine approach for the management of large and giant vestibular schwannomas. Otol Neurotol. 2002;23:224-227.
Misra BK. Surgery for giant acoustic neuroma: total excision at what cost. World Neurosurg. 2012;77:625-626.
Park CK, Jung HW, Kim JE, Son YJ, Paek SH, Kim DG. Therapeutic strategy for large vestibular schwannomas. J Neurooncol. 2006;77:167-171.
Patni AH, Kartush JM. Staged resection of large acoustic neuromas. Otolaryngol Head Neck Surg. 2005;132:11-19.
Pirouzmand F, Tator CH, Rutka J. Management of hydrocephalus associated with vestibular schwannoma and other cerebellopontine angle tumors. Neurosurgery. 2001;48:1246-1253; discussion 1253-1244.
Raslan AM, Liu JK, McMenomey SO, Delashaw JB Jr. Staged resection of large vestibular schwannomas. J Neurosurg. 2012;116:1126-1133.
Rath GP, Bithal PK, Chaturvedi A, Dash HH. Complications related to positioning in posterior fossa craniectomy. J Clin Neurosci. 2007;14:520-525.
Rhoton A. The cerebellopontine angle and posterior cranial fossa nerves by the retrosigmoid approach. Neurosurgery. 2000;47:93-129.
Rivas A, Boahene KD, Bravo HC, Tan M, Tamargo RJ, Francis HW. A model for early prediction of facial nerve recovery after vestibular schwannoma surgery. Otol Neurotol. 2011;32:826-833.
Roche PH, Ribeiro T, Fournier HD, Thomassin JM. Vestibular schwannomas: complications of microsurgery. Prog Neurol Surg. 2008;21:214-221.
Roser F, Tatagiba MS. The first 50s: can we achieve acceptable results in vestibular schwannoma surgery from the beginning? Acta Neurochir. 2010;152:1359-1365.
Samii M, Gerganov V, Samii A. Improved preservation of hearing and facial nerve function in vestibular schwannoma surgery via the retrosigmoid approach in a series of 200 patients. J Neurosurg. 2006;105:527-535.
Samii M, Gerganov VM, Samii A. Functional outcome after complete surgical removal of giant vestibular schwannomas. J Neurosurg. 2010;112:860-867.
Samii M, Matthies C. Management of 1000 vestibular schwannomas (acoustic neuromas): surgical management and results with an emphasis on complications and how to avoid them. Neurosurgery. 1997;40:11-21; discussion 21-13.
Sampath P, Rini D, Long DM. Microanatomical variations in the cerebellopontine angle associated with vestibular schwannomas (acoustic neuromas): a retrospective study of 1006 consecutive cases. J Neurosurg 92:70-78, 2000
Sanna M, Taibah A, Russo A, Falcioni M, Agarwal M. Perioperative complications in acoustic neuroma (vestibular schwannoma) surgery. Otol Neurotol 25:379-386, 2004
Sheptak PE, Jannetta PJ. The two-stage excision of huge acoustic neurinomas. J Neurosurg. 1979;51:37-41.
Silva J, Cerejo A, Duarte F, Silveira F, Vaz R. Surgical removal of giant acoustic neuromas. World Neurosurg. 2012;77:731-735.
Slattery WH, Schwartz MS, Fisher LM, Oppenheimer M. Acoustic neuroma surgical cost and outcome by hospital volume in California. Otolaryngol Head Neck Surg. 2004;130:726-735.
Sriskandan N, Connor SE. The role of radiology in the diagnosis and management of vestibular schwannoma. Clin Radiol. 2011;66:357-365.
Sugita K, Kobayashi S. Technical and instrumental improvements in the surgical treatment of acoustic neurinomas. J Neurosurg. 1982;57:747-752.
Tanaka Y, Kobayashi S, Hongo K, Tada T, Sato A, Takasuna H. Clinical and neuroimaging characteristics of hydrocephalus associated with vestibular schwannoma. J Neurosurg. 2003;98:1188-1193.
Tew JM., van Loveren HR, Keller JT. Atlas of Operative Microneurosurgery. Philadelphia: Saunders, 1994-2001.
Yates PD, Jackler RK, Satar B, Pitts LH, Oghalai JS. Is it worthwhile to attempt hearing preservation in larger acoustic neuromas? Otol Neurotol. 2003;24:460-464.
Please login to post a comment.









































Comments: